Quick Contact

MDR Article 117
MDR Article 117 of the Medical Device Regulation has amended Directive 2001/83/EC, Section 3.2, Point 12, mandating the involvement of a Notified Body for the issuance of a Notified Body Opinion (NBOp) for drug device combination products as a single integral device. Manufactures can use the Notified Body Opinion to include it in the Market Authorization Application to the European Medicines Agency.
The above article is applicable for devices where the manufacturer’s EU declaration of conformity and the relevant EU certificate issued by a notified body are not available.
Drug Device Combination Products
For a better understanding, drug device combination products, also known as integral products, are defined as follows:
- A medical device that incorporates a drug or medicinal substance as an integral part that, if used separately, is considered a drug or medicinal product and whose substance action is principal Examples; medicinal products with embedded sensors.
- A medical device intended to administer a drug or medicinal product that constitutes a single integral product intended exclusively for use in combination and which is not reusable Examples; single-use pre-filled syringes, drug-releasing intrauterine devices, and single-use pre-filled dry powder inhalers.
Non Drug Device Combination Products
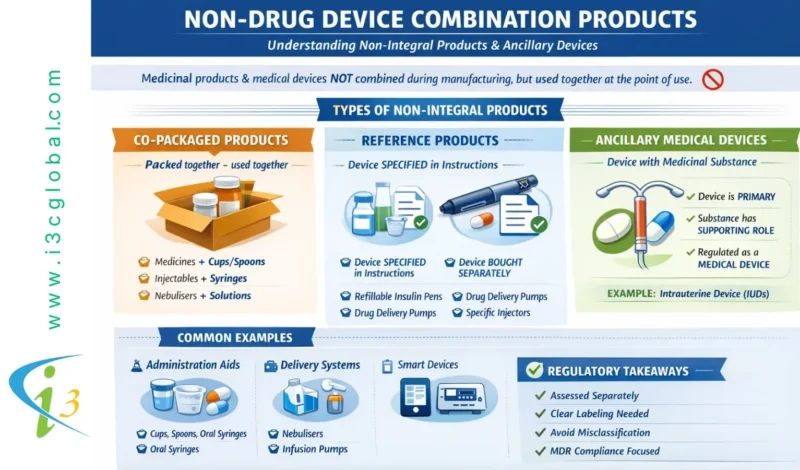
Medicinal and device components that are not physically integrated during manufacturing but are combined before human administration, so-called non-integral products, are not covered in drug-device combination products. Such types of products are detailed below.
- Co-packaged products – The medicinal product and the medical device are placed on the market and packed together into a single pack.
- Reference product – The medicinal product information (SmPC and/or package leaflet) is for a specific medical device that must be obtained separately.
A few examples of non-integral products are: cups, spoons and syringes for oral administration, refillable pens and injectors using cartridges, nebulisers, vaporisers, pumps for medicinal product delivery and electronic tablet dispensers.
- Ancillary Medical Devices – If the device incorporates a substance as an integral part that, if used separately, is considered a medicinal product, but the action of the substance is ancillary to that of the device. Examples are intrauterine device (IUD)
With the new MDR 2017/745, MDR Article 117 has brought significant impact. It now requires that the device portion of integral drug-device combination products must have an evaluation from a Notified Body to ensure compliance (NBOp)
MDR Article 117 Regulatory Framework
The new Medical Devices Regulation (MDR) 2017/745 (EU) sets stringent requirements on medical device manufacturers and notified bodies and broadens the scope to include the pharmaceutical industry about pharmaceuticals with a medical device as an integral component.
To determine whether a device part complies with the pertinent GSPR of Annex I, a notified body must be involved by MDR Article 117, which amends Directive 2001/83/EC, point 12 of Section 3.2.
Notified Body Opinion Report (NBOp)
A Notified Body conformity assessment of drug-device combination products coming under the preview of MDR Article 117 entails an examination of the technical documentation in relation to all applicable General Safety and Performance Requirements (GSPRs) stated in Annex I of the MDR.
After the technical documentation review is completed successfully, a Notified Body will issue an NBOp Report. This opinion can subsequently be included in the drug device combination product’s Marketing Authorization Application (MAA) submitted to a drug competent body such as the European Medicines Agency (EMA).
NBOp and Role of I3CGlobal Consultants
| No. | Consultant Activities |
|---|---|
| 1 | Find out if the product is a drug-device combination or an integral product.. |
| 2 | To find the main and ancillary components, identify the intended use and how it works. |
| 3 | Use your knowledge of pharmaceuticals and medical devices to come up with the regulatory strategy and route plan. |
| 4 | Check the medical device's risk classification and the rule and regulation number that apply. |
| 5 | Find the relevant harmonized and non-harmonized standards. |
| 6 | Follow the rules and do biological evaluations as needed. |
| 7 | Do a risk analysis and a benefit-risk assessment for the combined product. |
| 8 | Get ready for and look over the medical device's clinical evaluation. |
| 9 | Check the device's ease of use, cleanliness, packaging, and shipping needs. |
| 10 | Put together and organize documents in line with the General Safety and Performance Requirements (GSPR). |
| 11 | Help the manufacturer find and choose the right Notified Body. |
| 12 | Help with the application and submission for the Notified Body. |
| 13 | Make sure that communication is coordinated and that questions from the Notified Body are answered during the review of technical documentation. |
| 14 | During the Notified Body Opinion process, keep track of documents, make changes, and add new ones. |
I3CGLOBAL offers a team of QARA experts to assist with arranging Notified Body Opinion letters (NBOp), by preparing technical documentation, interacting with Notified Bodies, by correctly implementing requirements to ensure compliance with MDR Article 117.
Frequently Asked Questions
Which documents are required to be submitted to the Notified Body?
Timeline for Notified Body Opinion Letter
After providing the technical documentation in accordance with the GSPR, the Notified Body will conduct a preliminary review and accept the file. Typically, the full initial review of technical documentation takes 10 to 12 weeks. The timeline for the entire review process is determined by the compliance and objective evidence submitted against each section of the GSPR. The timeline is determined by the number of additional review rounds and questions posed by the notified body, as well as the manufacturer’s responsiveness in appropriately addressing the review points.