
EU IVDR CE Marking Process
Statement of Work (SOW) outlined below for the IVDR CE Marking process explains the activities, and responsibilities of consultants and CE applicants (Manufacturers) along with deliverables, for the on-time completion of technical documentation and submission to a Notified Body for obtaining CE marking under the European Union Medical Device Regulation (EU MDR 2017/746) for an In-Vitro-diagnostic device.
This Statement of Work (SOW) is designed to build awareness for newcomers applying for IVDR CE marking. Minor deviations in the steps may be necessary during actual documentation, depending on the device class and intended use.
| ACTIVITY | SCOPE OF I3CGLOBAL | SCOPE OF MANUFACTURER |
|---|---|---|
| PHASE 1 | ||
| Legal Address and Scope | Review and approve | Legal address Company Registration Certificate Incorporation Certificate |
| Review and approve | Critical outsourcing locations if any Address Confirmation QMS Implementation Status |
|
| PHASE 2 | ||
| Device Information | Request, review, guide and approve | Information on Previous Govt. Approvals if any |
| Device Models Confirmation | ||
| Generic name | ||
| GMDN Code & EMDN Codes | ||
| Trade Name/Brand Name | ||
| Device Variants Confirmation SKU (Stock keeping unit) | ||
| Previous and similar generations of the device | ||
| Overview of Identified Similar Devices | ||
| Medical Device Rationale | ||
| IVDR Codes | ||
| Device Description & Specifications | ||
| Review and Approval | Identifying key functional component Device Components Device Accessories Hardware/firmware/software |
|
| Request, review, guide and approve | Intended use | |
| Specification | ||
| Risk Classification, Rule, and Justification | ||
| Request, review, guide and approve | Software Level of Concern | |
| Indications | ||
| Principle of Assay Method (or) Principle of operation of the Instrument | ||
| Connections to the device | ||
| PHASE 3 | ||
| Confirmation of IVDR requirements and compliance issues | Identification of regulations | |
| Harmonized Standards | ||
| Non-Harmonized Standards | ||
| Device-Specific Standards and CLSI Guidelines | ||
| Other Applicable regulations if any | ||
| GSPR Checklist | ||
| Declaration of Conformity | ||
| Documentation of additional declarations | ||
| Review and Feedback |
Documentation as per ISO 13485
|
|
| Design Controls (Software) in line with ISO 62304 | Review and Feedback |
Documentation as per ISO 13485 / IEC 62304
|
| PHASE 4 | ||
| Risk Analysis & Usability | Development of Procedures and Templates and Implementation Guidance and coordination with manufacturer team |
Support and coordination with consultants in the risk management process.
|
| Risk Mitigation | ||
| Benefit-Risk Analysis | ||
| Risk Management File documentation | Review | |
| Usability engineering procedure and template | Support and suggestions | |
| Guidance, review and approval | Usability engineering report | |
| PHASE 5 | ||
|
Biocompatibility (It will be applicable based on the type and duration of contact with the human body) |
Biological Evaluation procedure and templates | |
| Identification of biocompatibility compliance requirements | ||
| Biocompatibility Plan | ||
| Guidance | Biocompatibility Test Reports | |
| Product Verification and Validation | Request, review, guide and approve | Information on the Performance of the device (Analytical Performance) |
| Request, review, guide and approve | Metrological Traceability | |
| Request, review, guide and approve | Chemical, Physical, and Biological Properties | |
| PHASE 6 | ||
| Production Controls | Request, review, guide and approve. | Process Flow chart |
| Critical Process Identification | ||
Critical Process Validation
|
||
Sterilization Validation
|
||
| Request, review, guide and approve. |
Clean Room Validation
|
|
| PHASE 7 | ||
| Device Labelling | Request, review, guide and approve. |
Device information Panel
|
| Instructions for Use/User Manual /Leaflet | IFU/User Manual | |
| Device Storage | Storage / Storage Temperatures | |
| Lifetime/Shelf life |
|
|
| Packaging Controls (Active, Non-Active Devices) |
Device Packing
|
|
| PHASE 8 | ||
| Quality Control | Request, review and approve. | Specifications of the finished device |
| Specification of Critical Raw Materials/Components | ||
| Quality Plan/Control Plan | ||
| Certificate of Analysis (COA) | ||
| Batch Release (Batch Manufacturing Record) | ||
| Request, review, guide and approve. | Performance Testing | |
| Review and approve. | Electrical Safety Testing | |
| PHASE 9 | ||
| Performance Evaluation |
Development of Quality System Procedure, if not available with the manufacturer
|
|
| Guidance and drafting of templates |
|
|
Develop a detailed Performance Evaluation Plan
|
|
|
| Performance Evaluation |
|
|
|
||
|
|
|
|
||
|
||
| Post-Market Surveillance and Vigilance Reporting |
|
|
| Post Market Performance Follow-up |
|
|
| PHASE 10 | ||
| Notified Body Submission, Review, Onsite Audit, and Issue of Certificate or NB Opinion Letter | Support in the Identification of Notified Body | |
| NB Application and Contact Signing | ||
| Technical Documentation Submission to NB | ||
| Answering initial NB review comments and resubmission of TDF | Support and suggestions | |
| Answering 2nd round NB review comments and resubmission of TDF | ||
| Onsite QMS Audit and closing of NC, s | ||
| Review draft IVDR CE Certificate | Confirm contents in the IVDR CE Certificate draft | |
| Accept IVDR CE Certificate (Soft/Hard) or Notified Body Opinion Letter | ||
EU IVDR CE Marking Process Flow Diagram
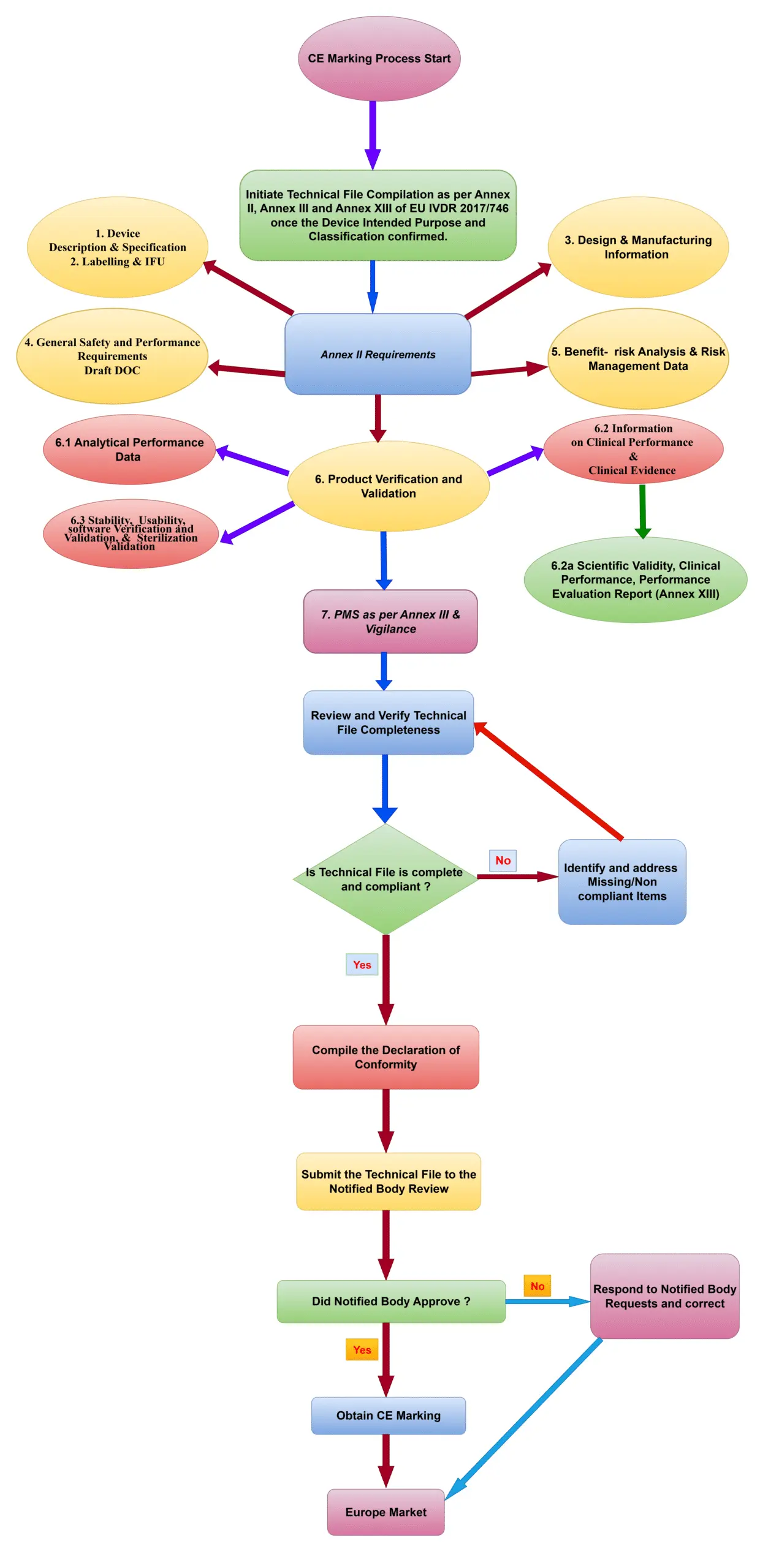
More details about Class A, Class A Sterile, Class B, Class C and Class D refer the respective pages.
