Quick Contact

Class 2a Medical Device
Class 2a Medical Device constitute medium-risk devices such as orthodontic wires, surgical gloves, lancets, etc. For Class 2a the declaration of conformity is backed up by notified body assessment post submission of technical documentation file. It holds a unique position within this classification system, embodying a delicate balance between innovation and patient safety.
It is a must to have experts on board who have previous experience with Class 2a Medical Device EU compliance and clinical evaluation for a successful outcome. Scope of I3CGLOBAL Class 2a consulting services are as follows:
- Class 2a Medical Device guidance
- Identification of standards
- Risk Analysis and Biological Evaluation
- Technical File Preparation
- Identifies test requirements and reviews the external reports
- Prepares Clinical Evaluation report as per MEDDEV 2.7/1 Rev 4.
- Prepare Periodic Safety Update Report
- Arrange Notified Body and coordinate with them till the issue of the CE Certificate
- Arrange EU Representative from the European Union
- EUDAMED Registration (SRN + Actor Registration)
- Arrange a Free Sale Certificate from the European Union
Do you need an email containing full details about Class 2a medical device within 2 minutes?
Class 2a Medical Device CE Conformity Assessment Route
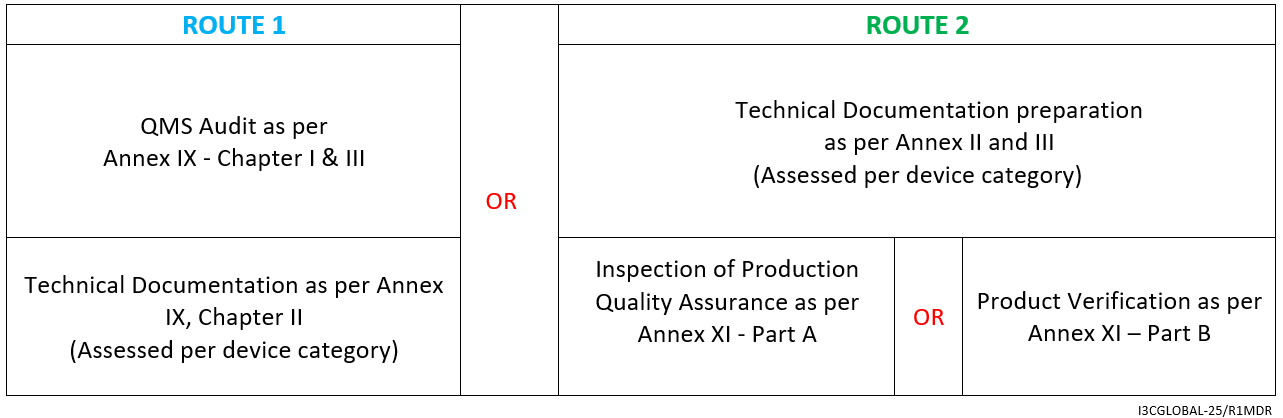
- Declaration of Conformity as per Annex IV
- Affixing the CE mark as per Annex V
Class 2a Medical Device Examples
Class 2a Medical Device encompass a wide range of medical technologies, often involving devices that come into contact with the body for diagnostic, therapeutic, or monitoring purposes. Some examples of Class 1a include:
1: Non-invasive Blood Pressure Monitors: Devices that measure a patient’s blood pressure without penetrating the skin fall under this category. These monitors are commonly used in clinics, hospitals, and home healthcare settings.
2: Infusion Pumps: Infusion pumps are used to deliver fluids, such as medications or nutrients, into a patient’s body in controlled amounts. They are vital in various medical treatments.
3: Contact Lenses: Non-corrective and corrective contact lenses for medical purposes fall under Class 2a medical device. These lenses can have various applications, such as therapeutic or cosmetic use.
4: Diagnostic Ultrasound Devices: Ultrasound devices used for diagnostic imaging purposes, such as obstetric ultrasound, are classified as Class 2a medical device. They are crucial tools in prenatal care and diagnostics.
5: Dental Filling Materials: Dental products like dental fillings and crowns that are intended to be placed within the oral cavity fall under this category.
Are you planning to obtain CE Certification through a Notified Body for Class 2a Medical Device or a System or a Kit? Contact us. We specialize in Documentation ensuring full compliance with all EU Regulatory requirements.
Frequently Asked Questions
Do I3CGLOBAL support with Notified Body slection
The CE Marking applicant/manufacturer is free to choose the notified body as long as the notified body is accredited by the competent authority in the relevant EEA state, Switzerland, or Turkey and has the relevant product group within its scope
What is the preferred route for Class 2a Medical Device CE Certification?
By chapters I and III of Annex IX OR compiling technical documentation by annexes II and III together with Conformity Assessment as specified in Section 10 or 18 of Annex XI
