FDA Sampling size guidance and plan provides an outline of how the testing is performed. It generally includes details like the standards used for the testing, sample size, test method, acceptance criteria, and data analysis plan.
The FDA sample size for performance testing, the bench is decided as per the standards- ANSI/ASQC Z1.4 or ISO 2859-1. The FDA Sampling Size for testing is selected based on the lot size and inspection level chosen. General Inspection level II is generally selected unless specified otherwise.
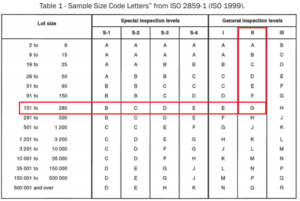
For example, from Table 1, for a lot size of 151 to 280 and general inspection level II, we select the sample size code “G”. There are different types of FDA sample plans- Single, double, and multiple. Also, the inspection level can be chosen as normal, reduced, or tightened.
Acceptance quality limit (AQL) is the worst tolerable quality level and indicates the maximum number of defective units permitted in a lot, beyond which the lot is rejected.
Based on the AQL selected and the sample size code, the sample size and the acceptance and rejection numbers are found out for the testing.
The sample size code is “G” (from table 1) and supposes the AQL is selected as 4%, from the table 2-A, the sample size is identified as 32 with the acceptance and rejection numbers as 3 and 4 respectively.
Even though the above method is generally used to find out the FDA Sampling Size for performance testing, some products have a fixed default number. For example, for the surgical masks (product code: FXX), FDA accepts 32 samples per lot as a default sample size for performance testing, irrespective of the lot size.
FDA recommends that the test be conducted on 3 non-consecutive lots or sufficient rationale be provided to justify the lot-to-lot performance of the surgical masks.
Information is published by Mrs. Neenu Jacob, Sr. Consultant (Medical Device Regulations) Email: nj@i3cglobal.in
