Quick Contact
Prepared by Experts. Low cost. Suitable for small and medium organizations.
Well Established Technologies (W.E.T.)
Navigating the European Medical Device Regulation (MDR 2017/745) is a complex journey, particularly when it comes to Clinical Evaluation Reports (CER). Whether your device is a “legacy” device or a cutting-edge innovation, the bar for clinical evidence has never been higher. At I3CGlobal, we specialise in turning regulatory hurdles into market advantages. We provide end-to-end clinical evaluation services that ensure your devices stay on the market and stay compliant.
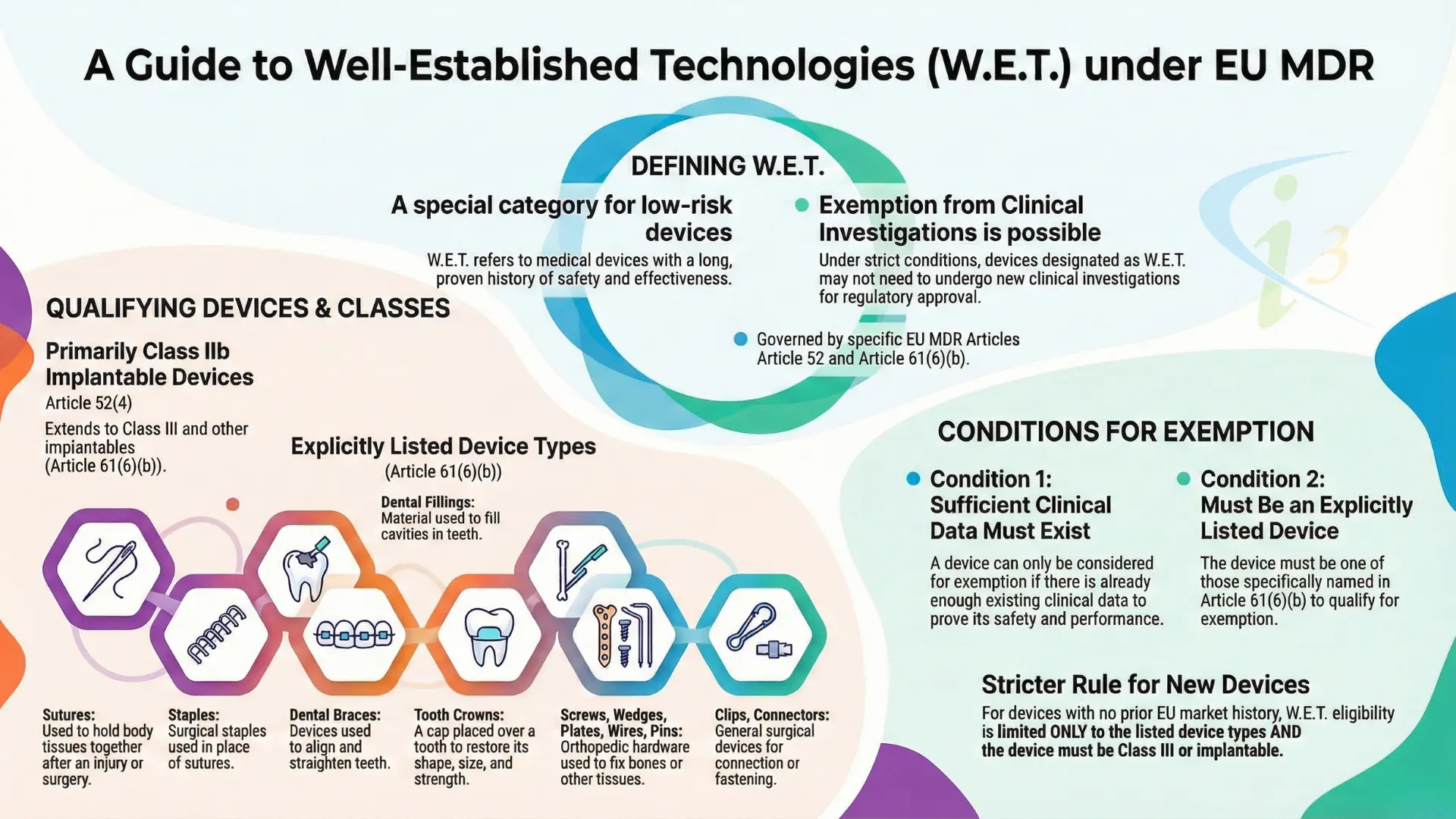
What are Well-Established Technologies (W.E.T.)?
Under the MDR, certain Class IIb and Class III implantable devices are categorised as Well-Established Technologies. These are devices that have been used clinically for decades with a known safety profile and high levels of predictability.
According to Article 61(6)(b), Sutures, Staples, Dental Fillings, Screws, Wedges, Plates, Wires, Pins, Clips, and Connectors may fall under this list. The W.E.T advantage is, if your device qualifies, you may be exempt from the requirement to perform new, dedicated clinical investigations, provided you have sufficient clinical data based on existing literature and equivalence.
The MDR has removed the “grandfathering” clause. Even if your device has been sold for 20 years, you must now prove its safety and performance through a rigorous CER (Clinical Evaluation Report). For W.E.T. devices, the challenge isn’t just having data it’s documenting it to the precise MDR requirements
⇒ Strict Equivalence: Proving your device is truly “equivalent” to the predicate in technical, biological, and clinical terms.
⇒ Continuous Updates: Clinical evaluation is no longer a one-time report; it is a living document tied to your Post-Market Clinical Follow-up (PMCF).
MDR Well Established Medical Technologies (W.E.T.) Device Classifications
| Device Category | Description of Use | MDR Article Reference | Device Class | Implantable Status | Eligibility Criteria |
|---|---|---|---|---|---|
| Sutures | Mechanical or structural role | Article 61(6)(b) | Class IIb or Class III | Yes | Sufficient clinical data available and listed in Art 61(6)(b) [1] |
| Staples | Mechanical or structural role | Article 61(6)(b) | Class IIb or Class III | Yes | Sufficient clinical data available and listed in Art 61(6)(b) [1] |
| Dental fillings | Mechanical or structural role | Article 61(6)(b) | Class IIb or Class III | Yes | Sufficient clinical data available and listed in Art 61(6)(b) [1] |
| Dental braces | Mechanical or structural role | Article 61(6)(b) | Class IIb or Class III | Yes | Sufficient clinical data available and listed in Art 61(6)(b) [1] |
| Tooth crowns | Mechanical or structural role | Article 61(6)(b) | Class IIb or Class III | Yes | Sufficient clinical data available and listed in Art 61(6)(b) [1] |
| Screws | Mechanical or structural role | Article 61(6)(b) | Class IIb or Class III | Yes | Sufficient clinical data available and listed in Art 61(6)(b) [1] |
| Wedges | Mechanical or structural role | Article 61(6)(b) | Class IIb or Class III | Yes | Sufficient clinical data available and listed in Art 61(6)(b) [1] |
| Plates | Mechanical or structural role | Article 61(6)(b) | Class IIb or Class III | Yes | Sufficient clinical data available and listed in Art 61(6)(b) [1] |
| Wires | Mechanical or structural role | Article 61(6)(b) | Class IIb or Class III | Yes | Sufficient clinical data available and listed in Art 61(6)(b) [1] |
| Pins | Mechanical or structural role | Article 61(6)(b) | Class IIb or Class III | Yes | Sufficient clinical data available and listed in Art 61(6)(b) [1] |
| Clips | Mechanical or structural role | Article 61(6)(b) | Class IIb or Class III | Yes | Sufficient clinical data available and listed in Art 61(6)(b) [1] |
| Connectors | Mechanical or structural role | Article 61(6)(b) | Class IIb or Class III | Yes | Sufficient clinical data available and listed in Art 61(6)(b) [1] |
Do you need an email containing more information about clinical evaluation? Privacy Policy >>
Facing Challenges? Partner with I3CGlobal
If your device is an innovative implantable or does not fall under the W.E.T. list provided in the above table, the path to a CE Mark is significantly steeper. In these cases, the MDR often mandates Clinical Investigations either retrospective or prospective. I3CGlobal ensures success for Non-W.E.T. devices by following the below steps
⇒ Identify exactly where your existing data falls short before you invest.
⇒ We help design and manage studies that satisfy NB’s while remaining cost-effective.
⇒ We explore every avenue to utilise data from similar devices to minimise new human studies.
Documentation is the bridge between your technology and the market. I3CGLOBAL provides the technical expertise to build that bridge. Our Specialized Services Include:
⇒ Comprehensive W.E.T. Documentation that meet Article 61(6)(b) requirements.
⇒ CER Writing & Remediation from scratch or fix “rejected” reports to get your compliance back on track.
⇒ We design Post-Market Clinical Follow-up plans that proactively gather the data you’ll need for your next audit.
⇒ Clinical Investigation Management and expert guidance for NB submissions.
Don’t let regulatory uncertainty stall your sales. Ensure your clinical documentation is as robust as your engineering.
Don’t Wait for a Notified Body Deficiency
The transition to MDR is not just a paperwork exercise, it is a critical business milestone. Whether you are dealing with Well-Established Technologies or complex innovative implants, I3CGlobal has the clinical and regulatory expertise to ensure your CERs are audit ready.
A rejected Clinical Evaluation Report can lead to months of lost sales and expensive remediation. Let our experts handle the heavy lifting of Article 61(6)(b) documentation and clinical data synthesis.
Frequently Asked Questions
Does being listed as a ``Well-Established Technology`` (W.E.T.) mean I am exempt from a Clinical Evaluation Report (CER)?
No. While Article 61(6)(b) provides an exemption from performing new clinical investigations (human trials) for specific devices like sutures, staples, and screws, it does not exempt you from the CER itself. You must still provide a robust Clinical Evaluation Report that appraises existing clinical data, demonstrates equivalence, and proves conformity with the MDR’s General Safety and Performance Requirements (GSPRs).
What happens if my device is equivalent to a W.E.T. device but is not on the specific Article 61 list?
If your device is an implantable or Class III device not explicitly listed in Article 61(6)(b) (e.g., a complex orthopedic implant), the MDR typically mandates a clinical investigation unless you can demonstrate equivalence to a device marketed by the same manufacturer. However, I3CGlobal specializes in finding pathways through rigorous Gap Analysis and “State-of-the-Art” (SOTA) literature reviews to determine if existing data can fulfill these requirements without the need for new, costly trials.
How often does W.E.T. documentation need to be updated?
Under the MDR, clinical evaluation is a continuous process, not a “one-and-done” report. For well-established technologies, the CER should typically be updated every 2 to 5 years, or sooner if new post-market surveillance (PMS) data or changes in the “State-of-the-Art” arise. I3CGlobal provides ongoing support to ensure your documentation remains live and compliant throughout the device’s lifecycle